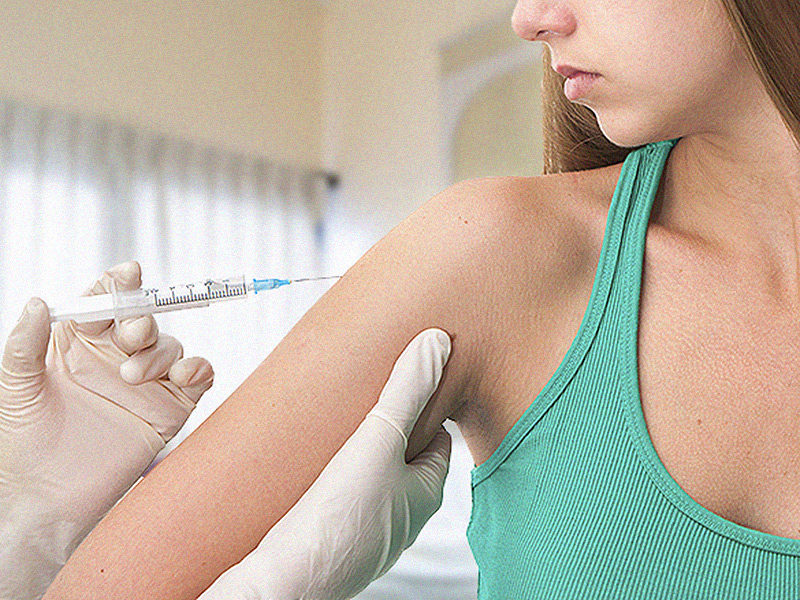
A handful of countries, like Denmark and Japan, remain skeptical about human papillomavirus (HPV) vaccines, even though the European Medicines Agency’s (EMA) Pharmacovigilance Risk Assessment Committee’s (PRAC) safety review in 2015 did not change the agency’s position that the benefits of HPV vaccinations outweigh the risks.
EMA Declares HPV Do Not Cause Two Painful Autoimmune Disorders
In July 2015, the PRAC in Europe launched a probe to investigate the safety of the three HPV vaccines available both in the United States and to countries in the European Union—Merck and Co.’s Gardasil and Gardasil 9 and GlaxoSmithKline’s Cervarix.
Continuing reports of several HPV-related adverse events prompted the investigation. The review focused on two inflammatory autoimmune conditions in particular: complex regional pain syndrome (CRPS),1 which causes sensory and motor dysfunction, including severe skin and bone pain; and postural orthostatic tachycardia syndrome (POTS)2 which increases heart rate abnormally and causes dizziness, fainting, chest pain, headache and weakness.
During routine vaccine safety monitoring, the PRAC had previously recorded reports of these two conditions in girls and women who had received HPV vaccines. Denmark specifically requested that the PRAC determine if there is a casual link between POTS and CRPS and HPV vaccinations.3
After interviewing HPV vaccine experts and evaluating published research, data from clinical trials, patient/provider reports and data supplied by Member States of the European Union, the PRAC concluded that the “available evidence does not support that CRPS and POTS are caused by HPV vaccines” and “therefore there is no reason to change the way the vaccines are used or amend the current product information.”
It is estimated that approximately 150 females aged 10 to 19 years out of every million may develop CRPS and/or POTS annually. Based on its review, the PRAC said the rates found among vaccinated females were consistent with the expected rates within this age group and noted that symptoms of CRPS and POTS overlap with other conditions, such as chronic fatigue syndrome.
The Committee for Medicinal Products for Human Use will consider the PRAC’s recommendation that government health officials make no change to the current HPV vaccine policy of giving all healthy girls and women a series of HPV vaccinations. The decision will eventually be adopted as the EMA’s final position.4
Some Doctors, Countries Remain Unconvinced
It was Denmark that initially requested the EMA review, and prominent physicians in that country are not convinced by the EMA’s conclusion that there should be no change to HPV vaccination policy. As a result, Denmark conducted and announced the results of its own independent investigation.
Dr. Jesper Mehlsen from Frederiksberg Hospital in Denmark explained that the EMA review was based on register studies with a focus on POTS and CRPS. Dr. Mehlsen pointed out that in some countries, POTS is not registered as a diagnosis.
Dr. Mehlsen, along with an independent researcher, Dr. Karsten Juhl Jorgensen, separately noted that many of the HPV vaccine adverse event reports come from the vaccine manufacturers themselves or those with substantial conflicts of interest, citing the potential for heavy bias. In fact, some of the EMA reviewers of HPV vaccine safety had been involved in the original HPV vaccine clinical trials and investigations. Therefore, they were, in effect, providing oversight on and judging themselves.
Denmark’s skepticism is backed by a number of case reports. Approximately 1,300 females have been referred to five specialist centers in Denmark with symptoms of CRPS or POTS. In particular, the Syncope Unit at Frederiksberg Hospital in Denmark reported 250 women and girls reporting these chronic symptoms within two months of vaccination.
Denmark’s Ministry of Health has awarded approximately $1 million for research by independent specialists treating girls with CRPS or POTS symptoms following HPV vaccinations. Results of the research are expected to be published during the first half of 2016.5
Other European Countries Struggling With HPV Vaccine Reactions
Other countries in Europe also are faced with addressing continuing reports of HPV vaccine injuries among their residents. In Spain, there had been 737 reports of HPV vaccine reactions, including three associated deaths, through January 2012. The creation of the Association of People Affected by HPV Vaccine (AAVP) was intended to address the issue of financial assistance for those suffering from HPV vaccine injury through lawsuits or government compensation.
The AAVP even requested that the Spanish Health Ministry ban HPV vaccines from the official vaccination schedule for the country.6 7 More recently, a public health inspector in Spain has launched a petition against the HPV vaccine that has accrued 35,000 signatures so far.
In 2014, France sent a similar petition to policymakers demanding further investigation into the safety of HPV vaccines with hundreds of thousands of signatures, including those from more than 700 doctors and 300 midwives.8
In early 2015, the United Kingdom established the Association of HPV Vaccine Injured Daughters (AHVID). In a survey among 94 AHVID members, 27 girls reported same-day reactions to the HPV vaccine, while 22 girls reported reactions within 7 days and 16 girls reported reactions with 30 days. Eight girls have a confirmed diagnosis of CRPS.9
Japan Continues Monitoring HPV Vaccine Reactions
After recording 1,968 adverse reactions—358 events deemed as serious—since the country began offering Gardasil vaccine to girls, Japan’s Vaccine Adverse Reactions Review Committee announced a moratorium on HPV vaccine use in June 2013. The Committee concluded that HPV vaccine should not be recommended for girls aged 12 to 16 years until safety concerns were fully addressed.10
Japan is considering overturning the decision for a moratorium on HPV vaccination, but is not yet convinced the vaccine is safe. In 2015, Japan’s Health Ministry surveyed 1,739 patients who received the HPV vaccine, revealing that 186 of them reported side effects, such as headaches, fatigue and muscle weakness. Further, 135 cases were severe enough to interfere with their daily activities.11 The government health agency determined an “undeniable causal relationship” for 11 of the cases and pay approximately $300 per month in government compensation is paid to each of the 11 HPV vaccine injured girls.12
In November, the private medical supply supervisory group, Medwatcher Japan, hosted a symposium titled, “Problems with the HPV Vaccine,” to further address reports that HPV vaccines are causing severe brain and immune system problems like POTS and CRPS.13
HPV Vaccine Injuries and Deaths Reported in US
In the United States, there have been continuing reports since Gardasil was licensed in 200614 that girls and women who get federally recommended HPV shots are having disabling reactions and some are dying following HPV vaccination.15 As of Oct. 14, 2015, there have been more than 41,000 Gardasil and Cervarix vaccine-related reactions, hospitalizations and injuries reported to the federally operated vaccine Adverse Events Reporting System (VAERS), including 226 deaths.16
The U.S. Centers for Disease Control (CDC) continues to maintain that HPV vaccines are safe.
References:
1 Goebel A, Blaes F. Complex regional pain syndrome, prototype of a novel kind of autoimmune disease. Autoimmunity Review 2013; 12(6): 682-686.
2 Hongliang L, Xichon Y et al. Autoimmune basis for postural tachycardia syndrome. J Am Heart Assoc 2014; 3(1).
3 European Medicines Agency. EMA to further clarify safety profile of human papillomavirus (HPV) vaccines. Press Release July 13, 2015.
4 European Medicines Agency. Review concludes evidence does not support that HPV vaccines cause CRPS or POTS. Press Release Nov. 5, 2015.
5 Chustecka Z. Chronic Symptoms After HPV Vaccination: Danes Start Study. Medscape Nov. 13, 2015.
6 Capilla A. AAVP asks Spanish Health Ministry to Ban HPV Vaccines. SaneVax Inc. Aug. 29, 2014.
7 Administration. Spain: AAVP responds to EMA conclusion on HPV vaccine safety. SaneVax Inc. Nov. 7, 2015.
8 Chustecka Z. Europe Again Says HPV Vax Safe, but Petition in Spain. Medscape Nov. 20, 2015.
9 See Footnote 3.
10 Judicial Watch. Japan Halts HPV Shot for Girls over Safety Issues. The Judicial Watch Blog Corruption Chronicles Oct. 1, 2013.
11 Kyodo Jiji. 10% of those harmed by cervical cancer vaccines failing to recover. The Japan Times Sept. 18, 2015.
12 Fifield A. The science doesn’t support them, but Japanese anti-vaxxers are winning on HPV. The Washington Post Nov. 10, 2015.
13 Symposium “Problems with the HPV Vaccine.” Medwatcher Japan Oct. 22, 2015.
14 NVIC. Merck’s Gardasil Not Proven Safe for Little Girls. June 26, 2006.
15 Gardasil and Unexplained Deaths.
16 MedAlerts.













11 Responses
“European Agency Declares HPV Vaccines Safe, But Denmark, Japan Skeptical”
Look at each member in the group and FOLLOW THE MONEY.
I would be willing to bet that the WHO has added their input which I’m sure influenced this ridiculous decision considering there is not ONE INDEPENDENT-GOLD STANDARD study or one shred of INDEPENDENT SCIENTIFIC DATA that confirms the HPV even causes cervical cancer. In fact the WHO along with the Task Force for Global Health has substantial input into all vaccine policies around the world. When it come to the HPV vaccines there is a clear conflict of interest:
Board Member Of The World Health Organization Is A Large Shareholder In Vaccine Company: edgytruth(dot)com/2015/03/09/board-member-of-the-world-health-organization-is-a-large-shareholder-in-csl-vaccine-company/.
There are no restrictions with regard to conflicts of interest for the employees of the CDC or for those of the FDA (Kuehn, 2010). Each employee of either agency is allowed to own stock in drug companies. There is of course the revolving door. The most blatant example of revolving door corruption may be the action of the former director of the CDC, Julie Gerberding. Gererding blocked the CDC’s planned retraction of their recommendation for HPV vaccine after significant numbers of girls had died or been rendered paralyzed by the HPV vaccine. Soon after blocking the retraction, Gerberding was offered and took a position at Merck, the manufacturer of Gardasil, the world’s best selling HPV vaccine, as head of Merck Vaccines Department (Reuters, 2009). It is a level of corruption that we would expect to see in only third world countries.
Kuehn, B. (Feb 3, 2010). Office of Inspector General: CDC Lax in Policing Advisors’ Conflicts of Interest. The Journal of the American Medical Association303. 5 412
There is no Cervical Cancer crisis or epidemic. The CDC & FDA allowed Merck to violate policy by Fast Tracking this drug. A drug is Fast Tracked if there is a WORLD wide imminent threat to the health of the world OF A CONTAGION. HPV vaccines DO NOT FALL INTO THAT CATEGORY THEREFORE THE CDC,FDA,WHO AND MERCK VIOLATED THAT POLICY.
This was illegal. The FDA requires new vaccines to undergo testing and a waiting period of 4 years. Gardasil was developed and on the market in 6 months, with FDA approval. The FDA approved the increase in Aluminum in Gardasil 9 without any scientific justification.
“10 December 2014: The FDA approved the use of a reportedly “new and improved” version of Gardasil, which will be marketed as Gardasil 9. According to the FDA approval letter, this action was taken without consultation with VRBPAC (the Vaccines and Related Biological Products Advisory Committee) which is responsible for reviewing and evaluating data concerning the safety, effectiveness, and appropriate use of vaccines and related biological products.
The FDA approval letter, signed by Marion Gruber, Director of Office of Vaccines Research and Review CBER, states the reason for bypassing the advice of VRBPAC writing:
”We did not refer your application to the Vaccines and Related Biological Products Advisory Committee because our review of information submitted in your BLA, including the clinical study design and trial results, did not raise concerns or controversial issues which would have benefited from an advisory committee discussion.”
So, the Office of Vaccines Research and Review, Center for Biologics Evaluation and Research (CBER) committee took it upon themselves to decide there were ”no concerns or controversial issues” regarding the approval of Gardasil 9?
This division of CBER decided there would be no benefit from ”an advisory committee discussion”?
According to their own mission statement, the FDA is ”responsible for protecting the public health by assuring the safety, efficacy and security of human and veterinary drugs, biological products, medical devices, our nation’s food supply, cosmetics, and products that emit radiation.”
The FDA, and all committees associated with the FDA, are public officials and therefore obliged to act in the public’s best interest particularly when it comes to health and safety issues.
Is bypassing advisory committee discussions regarding Gardasil 9’s potential safety and efficacy acting in the public’s best interest, or is it malfeasance, nonfeasance and/or misfeasance?” http://sanevax(dot)org/fda-approved-gardasil-9-malfeasance-or-stupidity/
Aluminum is a gene mutator. It is so toxic here is what Dr. Christoper Shaw had to say about it:
DR. CHRIS SHAW: ALUMINUM IN THE BODY – ONE MORE GIRL EXCERPTS: https://www.youtube(dot)com/watch?v=FfTo35UrFPA
-Merck’s vaccine trials used NON-INERT placebos for bogus “safety” results!
Please read the following:
-The origins of the HPV vaccine – A case of corruption in science: http://www.sott(dot)net/article/305663-SOTT-Exclusive-The-origins-of-the-HPV-vaccine-A-case-of-corruption-in-science303. 5 412.
-Merck’s Former Doctor Predicts that Gardasil will Become the Greatest Medical Scandal of All Time. http://healthimpactnews(dot)com/2014/mercks-former-doctor-predicts-that-gardasil-will-become-the-greatest-medical-scandal-of-all-time/.
-Gardasil Vaccine in Denmark: Serious Adverse Reactions Now Number 1 in 500 Girls. Articles found at Health Impact News- http://healthimpactnews.com/2015/tv2-denmark-documentary-on-hpv-vaccine-shows-lives-of-young-women-ruined/.
Sane Vax-http://sanevax(dot)org/; A very informative site with study results and stories from families whose children have been damaged by the HPV vaccine.
Cynthia A. Janak has actual test data(flawed) from the testing of the HPV vaccine and other information. http://www.renewamerica.com/columns/janak
Hi, I am Patrice cythia, from Houston, Texas, USA. I was living with HPV(human papilloma virus) for almost 2 years, i was not myself, my body was always inching with pains until i was on the internet searching for a remedy. So luckily, i was reading testimonials of clients who was cured of herpes by the amazing spell caster named Dr idedia, an ancient spell doctor from West Africa.I contacted him by email, and within 15 minutes he got back to me and ask of my details which i provide to him, he consulted his oracle and told me the solution to my problem and how to go about it, i did is he told me and about the two days i got the cure which he prepared and send to me through DHL service, i took the cure as instructed by him getting to two days i was cured, my HPV(human papilloma virus) was cured up. All thanks to the superb healing herbs of Dr idedia.
You too can get in contact with him today if you wanna be cured from HPV(human papilloma virus) you having by
email: [email protected] or what’sap +2349074505296
you can still get in touch with me to know more my cure, here is mine email [email protected]
visit his website https://dr-idedia-traditional-healing-home9.webnode.com/ Or
https://sites.google.com/view/dridediatraditionalhealinghome/home
his blog page https://dridediatraditionalhealinghome.blogspot.com/
https://patricecythia.blogspot.com/
Thank you so much great one.. I will continue to testify of your goodness to the universe.
NO VACCINE IS SAFE TODAY. MANY VACCINES ARE TAINTED BY THE UNDERWORLD GOVERNMENT THAT CONTROLS OUR GOVERNMENT. NEVER VACCINATE IF YOU HAVE ALREADY HAD YOUR CHILDHOOD VACCINES. VACCINES CAUSE AUTISM, SEIZURES, AUTO IMMUNE DISORDERS AND MUCH MORE. THEY EVEN CAUSE DEATH. WAKE UP PEOPLE! THEY WANT TO DEPOPULATE THE WORLD. THEY WANT TO KILL YOU OR MAKE YOU LEASS A THREAT WITH DISABILITIES. IT IS A VERY BIG CONSPIRACY. DO NOT TRUST THE GOVERNMENT OR THE FDA WHO SAYS THE VACCINES ARE OKAY! THEY ARE NOT!
Be very skeptical, just don’t do it. It’s just a money maker for the Pharmaceuticals and nothing more except issues for all those that get this drug, just DON’T GET It! Parents, don’t buy into the commercials. I have some nice land in Florida for sale as well. Do not buy into the crap you see on your TV, keep your kids safe.
EN FRANCE PLUS DE 700 CAS GRAVES AUSSI MAIS SILENCE RADIO
DE LA PART DES AUTORITÉS DE SANTE ACHETÉE PAR LE LABORATOIRE
https://www.change.org/p/marisol-touraine-stop-%C3%A0-la-g%C3%A9n%C3%A9ralisation-du-vaccin-gardasil
HPV vaccines have already been administered to some 70 million women worldwide, but their safety and effectiveness is questionable at best. The science used to justify their commercial approval simply doesn t stand up to scrutiny, and yet many countries throughout the world have adopted HPV vaccines as a preventative medicine for preventing cervical cancer.
INEFFICACY OF THE HPV VACCINE SEEN BY DOCTOR OF DEEP PERÚ
From its inception until the appearance of uterine cervical carcinoma (UCC) takes a average of 25-30 years; the research of this vaccine have begun in 2000. It is evident that the scientific efficacy of this new vaccine will be determined the years 2025 – 2030.
HPV not causes definitely the (CCU); at the onset of this disease involves multiple risk factors, including the suspected HPV, but scientifically is proven by epidemiology and statistics that the sex is what generates this disease. Nix in 100.000 nuns found not any UCC.
http://www.portalesmedicos.com/publicaciones/articles/1832/1/Epidemiolog.,.
There are not scientific researchs; stadistic, epidemiologic, citologic, histologic, colpocopic and clinic to demostrate that the HPV produce the cervical cance, are publishing. whitout scierntific sustentance.
To accept that a virus or a bacteria causes a infection disease must unfailingly fulfill the five Koch’s postulate
http://www.xatakaciencia.com/salud/los-postulados-de-koch
1 – The agent must be present in every case of the disease and absent from healthy.
2 – The agent must not appear in other diseases.
3 – The agent to be isolated in pure culture from disease lesions.
4 – The agent of causing disease in a susceptible animal being inoculated.
5 – The agent must again be isolated lesions in experimental animals.
http://es.scribd.com/doc/44558220/MICROBIOLOGIA-1
Consequently, HPV not fulfill not any principle of Koch’s postulates. by not meeting this postulate, that is accepted as dogma in medicine, scientifically we must be ensure that the HPV is not the causative agent to the UCC..
Until May 2013 Vaccine Adverse Event Reporting Syntem (VAERS) published that the vaccines against the HPV caused only in Unites States 138 muertes and 30020 adverse events; 947 disabled: 12 males, 924 females and 11sex unknown; 4050 advers graves: 106 males, 3883 females and 57 unknown sex; 527 abnormal PAP smears, 214 dysplasia cervical and cervical cancer 214. Vaccine Adverse Event Reporting System secure that only the !% to 10% are denounced https://dub104.mail.live.com/default.aspx#n=1521802 http://holyhormones.com/vaccinations/hpv-vaccine/hpv-vaccine-adverse-eve…
http://therefusers.com/?s=cervarix
The Vaccine efects advers reactions (VAERS) ensures that only complaint between 1% to 10% of the adverse effects produced by this evil vaccine;this figures shown are calculated according to the statements of the VAERS: to 10%.
http://www.noticiero.enkoria.com/2011/diez-menores-que-sufrieron-reaccio…
http://www.pop.org/content/merck-researcher-admits-gardasil-guards-again…
Dr. Harper, who contributed to the development of the vaccine by Merck, reports that the vaccine was not investigated in children under 15 years and the vaccine given to children under 11 years is a big public experiment.
http://offtheradar.co.nz/vaccines/53-researcher-diane-harper-blasts-gard…
The vaccine was approved to give girls uncontaminated with HPV, Dr. Howenstinc ensures that the women are vaccinated with HPV contaminated, have the possibility to acquire a 44.6% CCU
http://www.newswithviews.com / Howenstine/james170.htm.
Merck did not disclose that the vaccine was transgenic, the Sane Vax has discovered, which is transgenic because it has been found that the vaccine is contaminated with DNA recombinant vaccine Gardasil (DNArPVH) and has raised its concerns to the president of the FDA Margaret Hamburg. The FDA replied that the vaccine will not cause any damage transgenic
http://real-agenda.com/2011/09/16/vacuna-gardasil-contaminada-con-adn-re…
http://bolsonweb.com.ar/diariobolson/detalle.php?id_noticia=26075
A vaccinated child was ill with rheumatoid arthritis, which is an autoimmune disease. 24 hours after vaccination and found that the aluminum adhered to DNArPVH, two years after vaccination and in autopsy 6 months after death in a New Zeland girl Jazmine Renata which had recibed this deadly vaccines
http://www.mecfsforums.com/index.php?topic=9331.0
Management time to get market approval of a drug the FDA is at least three years, it is a drug for cancer 15 years, but the authorization Merck had only six months and the European Medicines Agency (EMA in English) only 9 months: To introduce the vaccine are using the marketing of fear
http://mujeresenaccion.over-blog.es/article-vph-la-vacuna-del-marketing-… http://mujeresenaccion.over-blog.es/article-vph-la-vacuna -of-marketing-of-fear-67210961.ht
HPV is ubiquitous; lives in wild and domestic animals, pollute us from birth, is on the doorknobs, on towels, on nails, on fomites, in gloves and specula of gynecologists,. sexual intercourse is not the only means of contamination.
http://spa.myhealthygood.com/cancer-cervical-vacuna-contra-el-vph/invest…
HPV also lives in the 400 nm outermost of our skin and mucous membranes. ,
If it live in our skin, our immune system produces cellular and humoral immunity is acquired or that our body is self vaccinatinge by PVHs living on our skin and mucous ..
http://www.conganat.org/seap/bibliografia/HPVToday/HPVToday007SEAP.pdf
The PVHs is not distributed uniformly worldwide. It has been found that in Canada HPV 18 only reaches 3%; is more often HPV 31, in my country Peru no studies have determined that HPV types predominate; Gardasil contains 225 mcg. aluminum and Cervarix 500 mcg, that produce the Alzheimer, Parkinson and autism, produce too neurotoxic and immune system disorders (Blaylock 2012) and polisorbato 80, a powerful contraceptive, that in experimental animals produces sterility, atrophy of the testicles and disturbance organic and funtional of the organs of the reproduction; is carcinogenic and mutagenic; also contains sodium borate considered poison unused in medicinal preparations (NLM)
http://www.telefonica.net/web2/paramahamsa/vacunaninosalerta.html http://detenganlavacuna.wordpress.com/2010/11/09/gardasil-cervarix/
Have been discovered to date 200 types of HPV; HPV is not infectious, contagious; the intercourse is not only that the persons is contaminated
http://quimicaclinicauv.blogspot.com/2006/08/virus-del-papiloma-humano.html http://www-lab.biomedicas.unam.mx/smpv/queeshpv.htm
On 22-11-2010 FDA approved Gardasil for males aged 9 to 26 to prevent warts and cancer to the anus, is overkill
http://real-agenda.com/2011/09/16/vacuna-gardasil-contaminada-con-adn-re…
http://salud.aollatino.com/2011/02/02/aprueba-fda-nueva-indicacion-vacun…
For the reasons from deep Peru Huancayo, I believe that this vaccine is a fraud?, a robbery?, a swindle?, a rough joke?, a crime?, a shame?, a scam?
The HPV is not scientifically proved for the moment that produce the UCC its effectiveness shall be verified just the years of 2025-2030.
Dr. Godofredo Arauzo
E mail: [email protected]
INEFFICACY OF THE HPV VACCINE SEEN BY DOCTOR OF DEEP PERÚ
Dr. Godofredo Arauzo
From its inception until the appearance of uterine cervical carcinoma (UCC) takes a average of 25-30 years; the research of this vaccine have begun in 2000. It is evident that the scientific efficacy of this new vaccine will be determined the years 2025 – 2030.
HPV not causes definitely the (CCU); at the onset of this disease involves multiple risk factors, including the suspected HPV, but scientifically is proven by epidemiology and statistics that the sex is what generates this disease. Nix in 100.000 nuns found not any UCC.
http://www.portalesmedicos.com/publicaciones/articles/1832/1/Epidemiolog.,.
There are not scientific researchs; stadistic, epidemiologic, citologic, histologic, colpocopic and clinic to demostrate that the HPV produce the cervical cance, are publishing. whitout scierntific sustentance.
To accept that a virus or a bacteria causes a infection disease must unfailingly fulfill the five Koch’s postulate
http://www.xatakaciencia.com/salud/los-postulados-de-koch
1 – The agent must be present in every case of the disease and absent from healthy.
2 – The agent must not appear in other diseases.
3 – The agent to be isolated in pure culture from disease lesions.
4 – The agent of causing disease in a susceptible animal being inoculated.
5 – The agent must again be isolated lesions in experimental animals.
http://es.scribd.com/doc/44558220/MICROBIOLOGIA-1
Consequently, HPV not fulfill not any principle of Koch’s postulates. by not meeting this postulate, that is accepted as dogma in medicine, scientifically we must be ensure that the HPV is not the causative agent to the UCC..
Until May 2013 Vaccine Adverse Event Reporting Syntem (VAERS) published that the vaccines against the HPV caused only in Unites States 138 muertes and 30020 adverse events; 947 disabled: 12 males, 924 females and 11sex unknown; 4050 advers graves: 106 males, 3883 females and 57 unknown sex; 527 abnormal PAP smears, 214 dysplasia cervical and cervical cancer 214. Vaccine Adverse Event Reporting System secure that only the !% to 10% are denounced https://dub104.mail.live.com/default.aspx#n=1521802 http://holyhormones.com/vaccinations/hpv-vaccine/hpv-vaccine-adverse-eve…
http://therefusers.com/?s=cervarix
The Vaccine efects advers reactions (VAERS) ensures that only complaint between 1% to 10% of the adverse effects produced by this evil vaccine;this figures shown are calculated according to the statements of the VAERS: to 10%.
http://www.noticiero.enkoria.com/2011/diez-menores-que-sufrieron-reaccio…
http://www.pop.org/content/merck-researcher-admits-gardasil-guards-again…
Dr. Harper, who contributed to the development of the vaccine by Merck, reports that the vaccine was not investigated in children under 15 years and the vaccine given to children under 11 years is a big public experiment.
http://offtheradar.co.nz/vaccines/53-researcher-diane-harper-blasts-gard…
The vaccine was approved to give girls uncontaminated with HPV, Dr. Howenstinc ensures that the women are vaccinated with HPV contaminated, have the possibility to acquire a 44.6% CCU
http://www.newswithviews.com / Howenstine/james170.htm.
Merck did not disclose that the vaccine was transgenic, the Sane Vax has discovered, which is transgenic because it has been found that the vaccine is contaminated with DNA recombinant vaccine Gardasil (DNArPVH) and has raised its concerns to the president of the FDA Margaret Hamburg. The FDA replied that the vaccine will not cause any damage transgenic
http://real-agenda.com/2011/09/16/vacuna-gardasil-contaminada-con-adn-re…
http://bolsonweb.com.ar/diariobolson/detalle.php?id_noticia=26075
A vaccinated child was ill with rheumatoid arthritis, which is an autoimmune disease. 24 hours after vaccination and found that the aluminum adhered to DNArPVH, two years after vaccination and in autopsy 6 months after death in a New Zeland girl Jazmine Renata which had recibed this deadly vaccines
http://www.mecfsforums.com/index.php?topic=9331.0
Management time to get market approval of a drug the FDA is at least three years, it is a drug for cancer 15 years, but the authorization Merck had only six months and the European Medicines Agency (EMA in English) only 9 months: To introduce the vaccine are using the marketing of fear
http://mujeresenaccion.over-blog.es/article-vph-la-vacuna-del-marketing-… http://mujeresenaccion.over-blog.es/article-vph-la-vacuna -of-marketing-of-fear-67210961.ht
HPV is ubiquitous; lives in wild and domestic animals, pollute us from birth, is on the doorknobs, on towels, on nails, on fomites, in gloves and specula of gynecologists,. sexual intercourse is not the only means of contamination.
http://spa.myhealthygood.com/cancer-cervical-vacuna-contra-el-vph/invest…
HPV also lives in the 400 nm outermost of our skin and mucous membranes. ,
If it live in our skin, our immune system produces cellular and humoral immunity is acquired or that our body is self vaccinatinge by PVHs living on our skin and mucous ..
http://www.conganat.org/seap/bibliografia/HPVToday/HPVToday007SEAP.pdf
The PVHs is not distributed uniformly worldwide. It has been found that in Canada HPV 18 only reaches 3%; is more often HPV 31, in my country Peru no studies have determined that HPV types predominate; Gardasil contains 225 mcg. aluminum and Cervarix 500 mcg, that produce the Alzheimer, Parkinson and autism, produce too neurotoxic and immune system disorders (Blaylock 2012) and polisorbato 80, a powerful contraceptive, that in experimental animals produces sterility, atrophy of the testicles and disturbance organic and funtional of the organs of the reproduction; is carcinogenic and mutagenic; also contains sodium borate considered poison unused in medicinal preparations (NLM)
http://www.telefonica.net/web2/paramahamsa/vacunaninosalerta.html http://detenganlavacuna.wordpress.com/2010/11/09/gardasil-cervarix/
Have been discovered to date 200 types of HPV; HPV is not infectious, contagious; the intercourse is not only that the persons is contaminated
http://quimicaclinicauv.blogspot.com/2006/08/virus-del-papiloma-humano.html http://www-lab.biomedicas.unam.mx/smpv/queeshpv.htm
On 22-11-2010 FDA approved Gardasil for males aged 9 to 26 to prevent warts and cancer to the anus, is overkill
http://real-agenda.com/2011/09/16/vacuna-gardasil-contaminada-con-adn-re…
http://salud.aollatino.com/2011/02/02/aprueba-fda-nueva-indicacion-vacun…
For the reasons from deep Peru Huancayo, I believe that this vaccine is a fraud?, a robbery?, a swindle?, a rough joke?, a crime?, a shame?, a scam?
The HPV is not scientifically proved for the moment that produce the UCC its effectiveness shall be verified just the years of 2025-2030.
Dr. Godofredo Arauzo
E mail: [email protected]
To whom it may concern:
Greetings from Global Vaccination 2017
The purpose of this letter is to solicit your gracious presence to be a Speaker at the upcoming “Global Vaccines and Vaccination Summit and B2B” to be held in Toronto during November 1-2, 2017, which is a globally illustrious annual conference.
The scientific program committee has established an instructive range of symposium, plenary lectures and poster communications.
Conference theme: “Discover and Network: The Trends in Vaccines and Global Health Issues”
An overview of the program can be found here.
Doctor I am delighted to notify you that as per your eminence in this field our organization has decided to waive off your registration amount and providing the best discount on accommodation that is USD 150, So, need to pay only USD 100 per night which includes breakfast, WIFI and we would provide lunch during conference days.
So, we request you to submit the abstract with title of talk and short biography.
Thank you and I would appreciate it if you could provide a response as soon as possible.
My best regards,
Maria Williams
Program Manager
There are several incidents supporting the case that HPV vaccination can cause severe chronic fatigue in high performance athletes.
It’s always difficult to prove such accusations but I know a young tennis player who developed light symptoms right after the first and severe symptoms after the second vaccination.
There are also reports of high performance athletes from Germany who experienced chronic fatigue after the vaccination.
Someone has to look into this!
My wife and I, have been infected with human papillomanvirus although i was the coursed i cheated on my wife it was when i contracted the virus for the pasted 3 years this has been a deeply situation so embarrassed every-time i looked at my self i just pissed off the virus was getting over my life. just to make the story short, few weeks ago i found a herbalist Dr. online who has cured hpv patients with 100% excellent result and i inquire the treatment me and my wife taking it for two weeks and we got cured i just want to use this medium to thanks Dr Agumba for his wonderful treatment he so-much saved us out from this virus his information been attached to my post you can reach out to him via
whatsapp num +2349032173881. or email [email protected]