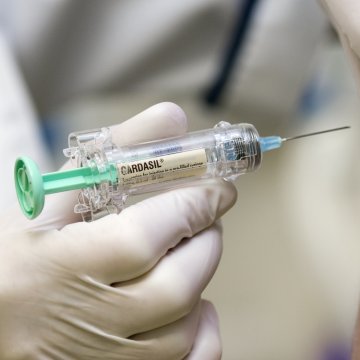
The American College of Pediatricians (ACP) released a statement last month expressing concerns about Merck and Co.’s human papillomavirus vaccine, Gardasil, and its correlation to premature ovarian failure, also known as premature menopause.
The ACP’s concerns are based on two case report series published since 2013 in which girls vaccinated with Gardasil developed premature menopause. Further, there have been 213 reports of premature menopause or amenorrhea (missing menstrual periods) recorded in the Vaccine Adverse Event Reporting System (VAERS) database since Gardasil’s approval in 2006.
Most of these reports were directly and solely linked to Gardasil. The ACP noted that during the pre-HPV vaccine period, there were no cases of premature menopause recorded in VAERS. The ACP also questioned the validity of Merck’s pre-licensure Gardasil clinical trials testing the vaccine’s safety because Merck did not use a true inactive placebo but, instead, used a bioactive “placebo” that contained two of the vaccine’s ingredients, one of which was aluminum.
Read the January 2016 press release from the ACP.













25 Responses
Have any safety studies been done on giving the vaccine to boys?? NY wants to start vaccinating all 6th graders with this vaccine. How can they do this without the proper safety studies being done (a placebo should be just saline, not polysorbate 80 mixed with aluminum)?
They never use a true placebo when testing any vaccines. They test against another vaccine or against an ingredient in the vaccine.
Hate to disappoint you Maria, but these vaccines get “fast track “approval with little or no testing. It is in your childs best interest to refuse these injections.
I thought this vaccine was just for girls. Its a vaccine for cervical cancer isn’t? Why would they give it to boys?
Where do you think women get HPV? Answer: men
If you know the cause, you should treat the cause.
Makes you wonder why there wasn’t a birth control pill for men!
They give it to boys because they say it helps prevent the boys from spreading it to the girls.
The Gardasil vaccine was fast tracked in the first place, so it’s not properly tested for anyone.
Can it be that an organization of pediatrician expresses any public reservation about a vaccination? This is shocking, shocking! Paired with local health departments, pediatricians have been at the vanguard of trying to dispel the “myth” of vaccination harm. Now, if only they can get the attention of their brethren, the American Academy of Pediatricians, who remain cloistered in their rigid insistence on vaccine safety, data be damned!
I agree, it is shocking, but encouraging! The only way they’re going to stop poisoning children is if the medical profession comes out en masse.
Let it be so that the AAP has their eyes opened too!! All of the children and adults damaged by this nonsense sickens me deeply…
I am so angry that my son and daughter-in-law forced my granddaughter to take this even after they called to ask what I thought of it and I said don’t.She is now experiencing severe cramps with her periods which she did not have before the vaccination.
I would like to see the sterilization statistics and death statistics published. To understand vaccination better try reading one of the whistleblowers Susan Humphries, “Dissolving Illusions” as a nurse myself there is the unwritten gag order in medicine where we are not permitted to speak about what vaccinations results are.
best book I have ever read, hands down
I am convinced that, among their other unholy agendas, depopulation is high on that list. Consider the many ways that we’re being poisoned:
chlorine and often fluoride in our drinking water along with a myriad of drug residues,
GMO “foods”, Monsanto’s poisonous glyphosphate in everything including remaining in our bodies,
fluoride in many prescription drugs, drugs for EVERYTHING,
the drugs they prescribe cause more life-threatening conditions and even death,
an agenda of forced vaccination with no way to exercise our God-given free choice,
smart meters everywhere, cell phone towers and the omnipresence of WiFi, “foods” that have been stripped of anything nourishing and laced with chemicals, lies from the medical mafia who insist that drugs are what we are lacking,
the continual geo-engineering that goes on in our once-deep blue skies that rains down aluminum, barium and only God knows what else,
autism that is now 1 in 50 where it used to be 1 in 10,000,
cognitive/behavioral issues in children are now the new norm,
diabetes is now the new normal, apparently and of course can only be “managed”
It is unending and evil. This world is being run by psychopaths who serve evil. Children are their biggest target; they are our future and the future looks bleak with what they’ve done to the little ones.
Amen! Great commentary, I couldn’t agree with you more. WE do all we can to protect our children, who by the way didn’t sign up for any of this. Parents need to wake up, speak up and exercise their (fleeting )rights on the aforementioned topics.
Nice to read remarks and opinions from like minded, rational thinking people.
Stay strong and keep up the fight
I agree with you 100% TO TELLTHETRUTH. It is a population control issue. All those smart, wealthy people think they are doing human beings a favor by deleting as many people as possible so food won’t run out. When men start thinking they are smarter than God, they are doomed to destroy everything. The only thing we can do is to try and educate as many people as possible about the dangers of vaccinations, prescription drugs, and all the chemicals in our food and water supply.
This really concerns me as I have 5 (soon to be 6 ☺) granddaughters. I firmly believe it is unnatural and unethical to force these or any vaccinations on everyone. Have we forgotten that we are biological creatures designed to thrive on this planet with the rest of nature. Our bodies are meant to build up natural immunity,and you need to experience some illness in life to do this.
We won’t have to worry about saving the planet for future generations if we continue to damage our children.
I believe many of the vaccines of this type are geared toward sterilization. Why are there so many couples unable to have a child? Infertility wasn’t an issue 25 years ago as it is now. This is how WHO, CDC, FDA, Big Pharma and Congress are coming together to control population all over the world.
Just like eboli and zika viruses they are saying they are now sexually transmitted. So, what better way to control population than to scare everyone all over the world they need to take a vaccination for all these viruses.
Parents and people need to wake up. Vaccinations are NOT immunization. Another avenue of population control is through our food using GMO’s and hybrid plants.
Just be aware folks, whenever the CDC, FDA, WHO, Big Pharma and Congress start telling us that vaccinations are mandatory, that is when we need to resist. All these “alerts” are nothing more than a lab experiment on us, the world lab, for all these organizations, and the sad thing is, Congress is clueless because they are getting their pockets lined by Big Pharma to pass laws, so that Big Pharma and doctors don’t get sued and to use the world as their laboratory to experiment with vaccines. The only way you get ANY compensation from damage from a vaccine is through arbitration with the drug company and a lawyer, and why is that? BIG PHARMA DOES NOT WANT THE PUBLIC KNOWING HOW DANGEROUS DRUGS AND VACCINES ARE, THEY KEEP IT QUIET THROUGH ARBITRATION, THE MAIN STREAM MEDIA WON’T COVER IT THE LAWSUITS AREN’T OUT INT HE PUBLIC FOR EVERYONE TO SEE. IT IS ALL HIDDEN WITHIN ARBITRATION, AND THE PARENTS ARE NOT ALLOWED TO GO PUBLIC, SO YOU SEE THIS IS HOW THEY KEEP THE DANGERS OF DRUGS AND VACCINATIONS HIDDEN.
Our children are their biggest laboratory!! Pray tell, why are so MANY precious children coming down with leukemias, blood dycrasias, brain tumors, neuromuscular issues, auto-immune issues? WHAT IS THE COMMON LINK IN ALL THESE? VACCINATIONS!!
Big Pharma uses third world countries first, to try out their vaccines, because their reasoning is that, overpopulated third world countries can afford to lose a few of their population. THIS IS DOWN RIGHT SICK AND DISGUSTING!! Big Pharma tested Gardasil in India on a group of girls, ages 8-16 and many of them died from the reaction from the Gardasil. THIS WAS NOT IN THE MEDIA, WHY? I Googled this and saw the story and was shocked!!
PLEASE PARENTS!!! EDUCATE YOURSELVES ABOUT THE DANGERS OF VACCINATIONS, AND BE READY TO CHALLENGE YOUR DOCTOR WITH FACTS, FIGURES, ARTICLES AND STATISTICS, TO EDUCATE HIM/HER!!! Doctors are clueless as well!! I saw a recent Vaccination schedule and it is up to 78 vaccinations from birth to 18 years of age. It used to be 69 vaccines from birth to 18 years of age. So, PLEASE THINK ABOUT THIS PARENTS!! YOUR PRECIOUS CHILDREN ARE BEING USED BY BIG PHARMA BECAUSE THEY CANNOT FIND WILLING VOLUNTEERS TO TEST THEIR VACCINES ON, SO WHAT BEST THING IS NEXT, THAN TO PASS LEGISLATION MAKING IT A LAW NO CHILD CAN ENTER SCHOOL UNLESS THEY HAVE HAD ALL THE VACCINATIONS?
Some thing needs to be done about this. I am so thankful for anti-vaccine organizations and doctors who are beginning to stand up.
If you will Google the news about the “anti-vaccine” doctors that are whistleblowers, you are going to find out how vicious Big Pharma is. Many of the whistleblowers have been murdered and it was reported as suicide or an “unfortunate” accident. These doctors who blew the whistle knew they were at risk, but they chose to have a conscience and speak up.
CE VACCIN A DÉTRUIT LA VIE DE MA FILLE
THIS VACCINE DESTROYED THE LIFE OF MY DAUGHTER
SIGN OURS PETITION TO HAVE THE REAL TRUTH ON THIS GARDASIL
https://www.change.org/p/marisol-touraine-stop-%C3%A0-la-g%C3%A9n%C3%A9ralisation-du-vaccin-gardasil
Approved in 1996, the Gardasil vaccination I got in 2007 GAVE ME & MY WHOLE BODY HARD-TO-ERADICATE WARTS. When I said that OUT-LOUD to my OB-GYN, she showed shock at my knowing that it Gardasil IS the CAUSE of the aggressive form of warts my body has!!!
OTC treatments do NOT work.
After 8 months of Dry Ice, these treatments do NOT ERADICATE HARD-TO-ERADICATE WARTS that disguise themselves as “partially healed” only to return again months or even years later.
These HARD-TO-ERADICATE WARTS MUST NEEDS BE SURGICALLY REMOVED . . .
. . . OUCH!!!!!!!!!!!!!!!!! 🙁
As a chiropractor, I have always taken issue with mandatory vaccination. The MD’s jumped on the Gardisil bandwagon so quickly and there was no time for any long term studies. I am so glad I didn’t cave to immense pressure to have my daughter or son vaccinated. Yet another cash cow for the drug companies goes awry! I can’t say that I’m surprised.
I feel that I have a right to be wary of vaccines since one of the earliest, the polio vaccine, was contaminated with simian viruses, the 40th one (SV40) is a cancer causing virus. These viruses were only found in monkeys until the polio vaccine introduced them into the population. They went undetected for 8 years. Now it has been shown that asbestos exposure, and having the SV40 virus is implicated in the rise of mesothelioma. So just for this example alone, I feel I would be foolish to not question vaccines! Also, before the polio epidemics of the 20th century, polio was not known as a paralyzing disease. Polio naturally infected people usually without symptoms whatsoever. If polio had been around forever, and did not cause major problems, what could’ve been causing all the paralysis and deaths? DDT was commonly used on food, in kitchens, even in children’s wallpaper.(look up DDT is good for meee). Children used to run through the DDT truck mists playing in the fog. People were using DDT routinely in the thought that bug free was healthier.
Also, physicians were putting children in casts, making them unable to use their muscles. Sister Kinney from Australia came along and said that that method was all wrong. She was highly influential in getting medicine to change their practices.
There are many examples of medical dogma that turned out to be dead wrong. From Semmelweis being ostracized for recommending hand washing prior to general acceptance of germ theory, to the recommendation of hydrogenated fats for heart health over natural fats, medicine has many times gotten it wrong. I appreciate all that modern medicine can offer as far as trauma care, but as for general health and longevity, they are far behind. For example, strong chemotherapy continues to be used for pancreatic cancer, even though outcomes with conventional therapies are bleak. Google bitter mellon for pancreatic cancer. Google pubmed resveratrol multiple myeloma. Pubmed just about any disease and curcumin. The cure for malaria is an herb, the Nobel prize was recently won showing that, but it’s not new knowledge. In a nutshell, nature knows what it’s doing.
Hi Kim, I agree with you on most of your post. Don’t be fooled by the claims that DDT killed animals, harmed the environment and caused cancer. There is a documentary (no longer free to view unfortunately, called 3 Billion and Counting). This documentary (if you get a chance to see it) will change everything you thought you knew about DDT. I know it did for me. I, like most of the population, grew up believing DDT was a heinous pesticide that did great damage to the environment. Just as with any government program or claim, be weary of the “evidence” and follow the money. The money trail always leads to the truth.
If you don’t get a chance to view the documentary be sure to look up Dr. Rutledge and investigate for yourself. I highly recommend seeing the video. It’s quite lengthy but one of the most eye opening docu’s I’ve seen.
Regards,
Candace
Although I generally agree with everything that has been said in the article, using the American College of Pediatrics as a source is less than confidence building. This particular Association has somewhat of a controversial reputation citing articles like the following: http://www.acpeds.org/same-sex-marriage-not-best-for-children. Having shared this post on Facebook without doing the due diligence of checking the source and relying on NVIC’s credibility (my bad) I was ridiculed by some of my colleagues for citing this source.
INEFFICACY OF THE HPV VACCINE SEEN BY DOCTOR OF DEEP PERÚ
From its inception until the appearance of uterine cervical carcinoma (UCC) takes a average of 25-30 years; the research of this vaccine have begun in 2000. It is evident that the scientific efficacy of this new vaccine will be determined the years 2025 – 2030.
HPV not causes definitely the (CCU); at the onset of this disease involves multiple risk factors, including the suspected HPV, but scientifically is proven by epidemiology and statistics that the sex is what generates this disease. Nix in 100.000 nuns found not any UCC.
http://www.portalesmedicos.com/publicaciones/articles/1832/1/Epidemiolog.,.
There are not scientific researchs; stadistic, epidemiologic, citologic, histologic, colpocopic and clinic to demostrate that the HPV produce the cervical cance, are publishing. whitout scierntific sustentance.
To accept that a virus or a bacteria causes a infection disease must unfailingly fulfill the five Koch’s postulate
http://www.xatakaciencia.com/salud/los-postulados-de-koch
1 – The agent must be present in every case of the disease and absent from healthy.
2 – The agent must not appear in other diseases.
3 – The agent to be isolated in pure culture from disease lesions.
4 – The agent of causing disease in a susceptible animal being inoculated.
5 – The agent must again be isolated lesions in experimental animals.
http://es.scribd.com/doc/44558220/MICROBIOLOGIA-1
Consequently, HPV not fulfill not any principle of Koch’s postulates. by not meeting this postulate, that is accepted as dogma in medicine, scientifically we must be ensure that the HPV is not the causative agent to the UCC..
Until May 2013 Vaccine Adverse Event Reporting Syntem (VAERS) published that the vaccines against the HPV caused only in Unites States 138 muertes and 30020 adverse events; 947 disabled: 12 males, 924 females and 11sex unknown; 4050 advers graves: 106 males, 3883 females and 57 unknown sex; 527 abnormal PAP smears, 214 dysplasia cervical and cervical cancer 214. Vaccine Adverse Event Reporting System secure that only the !% to 10% are denounced https://dub104.mail.live.com/default.aspx#n=1521802 http://holyhormones.com/vaccinations/hpv-vaccine/hpv-vaccine-adverse-eve…
http://therefusers.com/?s=cervarix
The Vaccine efects advers reactions (VAERS) ensures that only complaint between 1% to 10% of the adverse effects produced by this evil vaccine;this figures shown are calculated according to the statements of the VAERS: to 10%.
http://www.noticiero.enkoria.com/2011/diez-menores-que-sufrieron-reaccio…
http://www.pop.org/content/merck-researcher-admits-gardasil-guards-again…
Dr. Harper, who contributed to the development of the vaccine by Merck, reports that the vaccine was not investigated in children under 15 years and the vaccine given to children under 11 years is a big public experiment.
http://offtheradar.co.nz/vaccines/53-researcher-diane-harper-blasts-gard…
The vaccine was approved to give girls uncontaminated with HPV, Dr. Howenstinc ensures that the women are vaccinated with HPV contaminated, have the possibility to acquire a 44.6% CCU
http://www.newswithviews.com / Howenstine/james170.htm.
Merck did not disclose that the vaccine was transgenic, the Sane Vax has discovered, which is transgenic because it has been found that the vaccine is contaminated with DNA recombinant vaccine Gardasil (DNArPVH) and has raised its concerns to the president of the FDA Margaret Hamburg. The FDA replied that the vaccine will not cause any damage transgenic
http://real-agenda.com/2011/09/16/vacuna-gardasil-contaminada-con-adn-re…
http://bolsonweb.com.ar/diariobolson/detalle.php?id_noticia=26075
A vaccinated child was ill with rheumatoid arthritis, which is an autoimmune disease. 24 hours after vaccination and found that the aluminum adhered to DNArPVH, two years after vaccination and in autopsy 6 months after death in a New Zeland girl Jazmine Renata which had recibed this deadly vaccines
http://www.mecfsforums.com/index.php?topic=9331.0
Management time to get market approval of a drug the FDA is at least three years, it is a drug for cancer 15 years, but the authorization Merck had only six months and the European Medicines Agency (EMA in English) only 9 months: To introduce the vaccine are using the marketing of fear
http://mujeresenaccion.over-blog.es/article-vph-la-vacuna-del-marketing-… http://mujeresenaccion.over-blog.es/article-vph-la-vacuna -of-marketing-of-fear-67210961.ht
HPV is ubiquitous; lives in wild and domestic animals, pollute us from birth, is on the doorknobs, on towels, on nails, on fomites, in gloves and specula of gynecologists,. sexual intercourse is not the only means of contamination.
http://spa.myhealthygood.com/cancer-cervical-vacuna-contra-el-vph/invest…
HPV also lives in the 400 nm outermost of our skin and mucous membranes. ,
If it live in our skin, our immune system produces cellular and humoral immunity is acquired or that our body is self vaccinatinge by PVHs living on our skin and mucous ..
http://www.conganat.org/seap/bibliografia/HPVToday/HPVToday007SEAP.pdf
The PVHs is not distributed uniformly worldwide. It has been found that in Canada HPV 18 only reaches 3%; is more often HPV 31, in my country Peru no studies have determined that HPV types predominate; Gardasil contains 225 mcg. aluminum and Cervarix 500 mcg, that produce the Alzheimer, Parkinson and autism, produce too neurotoxic and immune system disorders (Blaylock 2012) and polisorbato 80, a powerful contraceptive, that in experimental animals produces sterility, atrophy of the testicles and disturbance organic and funtional of the organs of the reproduction; is carcinogenic and mutagenic; also contains sodium borate considered poison unused in medicinal preparations (NLM)
http://www.telefonica.net/web2/paramahamsa/vacunaninosalerta.html http://detenganlavacuna.wordpress.com/2010/11/09/gardasil-cervarix/
Have been discovered to date 200 types of HPV; HPV is not infectious, contagious; the intercourse is not only that the persons is contaminated
http://quimicaclinicauv.blogspot.com/2006/08/virus-del-papiloma-humano.html http://www-lab.biomedicas.unam.mx/smpv/queeshpv.htm
On 22-11-2010 FDA approved Gardasil for males aged 9 to 26 to prevent warts and cancer to the anus, is overkill
http://real-agenda.com/2011/09/16/vacuna-gardasil-contaminada-con-adn-re…
http://salud.aollatino.com/2011/02/02/aprueba-fda-nueva-indicacion-vacun…
For the reasons from deep Peru Huancayo, I believe that this vaccine is a fraud?, a robbery?, a swindle?, a rough joke?, a crime?, a shame?, a scam?
The HPV is not scientifically proved for the moment that produce the UCC its effectiveness shall be verified just the years of 2025-2030.
Dr. Godofredo Arauzo
E mail: [email protected]