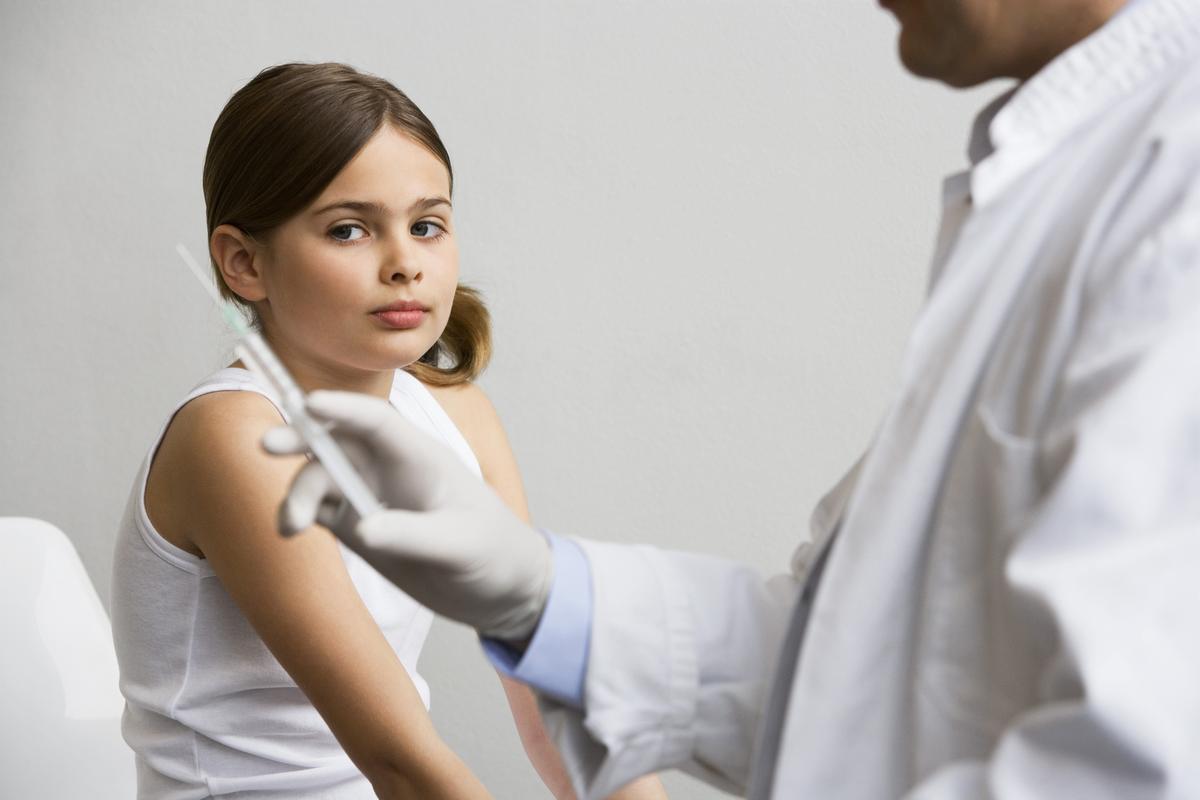
U.S. government health officials continue to push hard for a countrywide mandate of human papillomavirus (HPV) vaccination. They maintain that the cost-benefit ratio of the new nonavalent Gardasil vaccine, which contains nine HPV strains, the ticket to succeeding in their efforts to get every girl and boy in America to take the vaccine.
Nonavalent Vaccine Covers More Disease Strains
In 2006, a 4-strain version of Merck’s Gardasil vaccine was the first HPV vaccine to be fast tracked to licensure in the U.S. and it contained two HPV strains associated with cervical cancer and two HPV strains associated with genital warts.1
In 2014, the U.S. Food and Drug Administration (FDA) approved Merck’s nonavalent vaccine that targets five more types of sexually transmitted HPV strains associated with cancers.2 The nine-strain vaccine is now approved for use in females and males aged 9 to 26 years and is administered in three separate shots.3
In its February 2015 meeting, the U.S. Center for Disease and Prevention’s (CDC) Advisory Committee on Immunization Practices (ACIP) recommended the nonavalent Gardasil vaccine as an option for routine HPV vaccination.
With 30 strains of HPV that specifically affect at least half of sexually active females and males, the nine HPV Gardasil vaccine may seem to bump up the vaccine’s protection. However, the additional five HPV strains targeted by the nonavalent vaccine are related more to HPV-associated cancers in females rather than cancers in males. In fact, ACIP concluded that the additional protection from the nonavalent vaccine will mostly benefit females.4
According to a report by the Henry J. Kaiser Family Foundation, the five additional HPV strains are associated with approximately 20% of all cervical cancer cases.5
Government Continues Push for Nationwide Mandate
A 2016 study published in the Proceedings of the National Academy of Science reported a positive cost/benefit ratio for universal use of the nonavalent version of the HPV vaccine. Government health officials are likely to use this study as an impetus for even more strongly promoting a national HPV vaccine mandate for all children.
Yale University researchers created a model of HPV transmission, cervical cancer incidence and HPV vaccination rates, taking into account state-specific demographics, sexual behavior and migratory patterns. They used their model to “test” the cost-effectiveness by assuming that children in all states would get the nonavalent HPV vaccine.
At the national level, the researchers concluded that the nonavalent version achieved cost-effectiveness when compared to the bivalent and quadrivalent versions.6
In terms of health impact, the study authors predicted that switching to the nine-strain version would decrease cervical cancer incidence by 73%, compared with 63% with older vaccines that contained fewer HPV strains. The model also predicted a reduction in deaths due to HPV associated cancers by 49% versus 43%.7
The researchers concluded that switching to the nonavalent version would have the same impact as raising the rates of vaccination by 11%, but would cost $3 billion less. They estimated that nationwide use of the new Gardasil vaccine would translate to a state-specific health savings per capita of $4.40.8
“[I]f policies promoting nonavalent vaccine implementation and expansion of coverage are coordinated across multiple states, all states benefit both in health and in economic terms,” the researchers wrote.9
In a Conflict of interest statement at the end of the published study, the lead author and a co-author reported they have consulted for and received research funding from Merck and Sanofi Pasteur. Another co-author has consulted for and received research funding from GlaxoSmithKline, which markets the HPV vaccine, Cervarix, in the U.S.
Gardasil 9 cost roughly between $140 to $180 per dose, compared with the $130 to $150 range for the older quadrivalent Gardasil.10 Insurance companies are expected to start covering Gardasil 9 in January 2017.
Low Vaccination Rates Still Trending
Though government efforts seem to be ramping up to secure a national mandate, HPV vaccination rates remain low.
According to the CDC, in 2014, receipt of all three doses among girls aged 13 to 17 years was 39.7%. For teenage boys, it was only 21.6%.
Half of the states in the U.S. have proposed programs to increase HPV vaccination. So far, only three states have enacted HPV vaccine mandates for school attendance (Washington, DC, Virginia, Rhode Island), although 25 states and territories have proposed legislation that has not passed yet.11
In Virginia and Washington, DC, currently parents can opt to decline the vaccine for their children.12
References:
1 Rabin R. A New Vaccine for Girls, But Should It Be Compulsory? The New York Times July 18, 2006.
2 U.S. Food and Drug Administration. FDA approves Gardasil 9 for prevention of certain cancers caused by five additional types of HPV. Press Release Dec. 10, 2014.
3 Merck and Co. FDA Approves Expanded Age Indication for Gardasil 9 in Males. Press Release Dec. 15, 2015.
4 U.S. Centers for Disease Control and Prevention. Use of 9-Valent Human Papillomavirus (HPV) Vaccine: Updated HPV Vaccination Recommendations of the Advisory Committee on Immunization Practices. Morbidity and Mortality Weekly Report Mar. 27, 2015.
5 The Henry J. Kaiser Family Foundation. The HPV Vaccine: Access and Use in the U.S. Sept. 3, 2015.
6 Durham DP, Ndeffo-Mbah ML, Skrip LA, et al. National- and state-level impact and cost-effectiveness of nonavalent HPV vaccination in the United States. Proceedings of the National Academy of Sciences 2016.
7 Yale University. New HPV vaccine could curb cervical cancers and health costs if adopted widely. Science Daily Apr. 18, 2016.
8 Saxena R. National HPV vaccination program would provide big benefits. Ars Technica Apr. 24, 2016.
9 See Footnote 5.
10 CDC. CDC Vaccine Price List. Updated May 2, 2016.
11 National Conference of State Legislatures. HPV Vaccine Policies. Accessed May 16, 2016.
12 National Vaccine Information Center. State Law and Vaccine Requirements. NVIC.org.












22 Responses
What about all the deaths and disabilities< they forgot to factor this in? And the fact that cervical cancer rates ROSE in Denmark, in girls given the vaccine, How CORRUPT CAN CDC GET?
The CDC is part of the fed gov. Those at the top in all gov agencies are psychopaths bent on control no matter the “cost” to the populace. Unfortunately, most of those hired to perform the daily tasks are just not that bright to question what they are doing. Plus one hand never knows what the other is doing.
I’ve been in alternative health care for 46 years. I have personally witnessed docs being persecuted and disbarred for using alternative methods to treat disease. Recently, 11 doctors were found dead – all suspicious deaths but of course the media calling them “accidents” or suicides. Most were healing autistic children and other conditions without drugs and drawing more media attention to this fact.
The people at the top are cold-blooded killers – that’s how corrupt the CDC, FDA, AMA and other agencies and organization are. The media is controlled by the very same organizations so you will never hear about that side of things. Strength comes in numbers, or so they say. Too few people truly know about these things because they sit around watching TV, playing video games or listening to NPR – yep even that radio show is quiet about it all.
Until people get off their b*tts and research this, we will continue on this dark road to hell.
Actually its 51 deaths now since fathers day 2015. There were just 2 more on Thursday. You can link to http://www.Excaliburreport.com for more info.
Murderously corrupt. Parents must wake up to this fraud. Everyone should stop all vaccinations now.
We live in Rhode Island, and before my son can start 7th grade he will be required to get this vaccine. This scares the hell out of me. The only two exemptions afforded to us are medical (for which we must prove a valid contraindication, signed off on by his doctor) or religious (which states that if an outbreak should occur, my son would be expelled from school until he gets vaccinated). I don’t know what to do.
It’s insane to mandated this vaccine for school attendance. It is not a communicable disease. It is not airborne. It is a STD!!! I have 3 boys they will not be getting this vaccine or any other vaccine. B
If you lived in GA I would provide the exemption. Perhaps you can find an enlightened doctor where you live.
TeeJae, I have not allowed doctors to give any vaccines to my 4 children. They are all teens now… not once have they ever had to miss out on school because of an outbreak. And even if they did, what a small price to pay anyway, right?! Its just another form of fear mongering to those of us who want to choose what is best for our children. Follow what your gut is telling you to do in the best interest of YOUR children. Good luck!
Get in touch with this group. They are fighting it and you will all be stronger together. Fight it on a national level also. https://www.facebook.com/groups/RIagainstHPVmandate/?fref=nf
Just file for religious exemption. There are no outbreaks. Don’t chance the health of your child. The HPV vaccines is thought to actually make individuals more susceptible to the other strains, and that is if you are lucky and don’t get injured by it.
CIRCUMCISION IS A CRIME AGAINST BOYS AND SOMETIMES GIRLS AND VACCINATIONS ARE ALSO.
PARENTS ARE KEPT TOO BUSY TO INVESTIGATE SO THEY ARE FORCED TO BELIEVE THE CORRUPT CDC AND THE DOCTORS THAT RUN THAT OPERATION.
LOOK AT ALL THE CRAP ABOUT THE SO-CALLED ZICA VIRUS WHERE THEY ARE RUSHING A VACCINE THROUGH BEFORE THE ‘EPIDEMIC’ DISAPPEARS LIKE EBOLA ETC.
ALL THOSE TROPICAL DISEASES ARE DUE TO THE INABILITY OF AN INDIVIDUAL TO HANDLE THE WATER THEY BREATHE IN. YELLOW FEVER, DENGUE FEVER, MAL-ARIA ( BAD AIR) ETC. THESE DRUG PUSHERS ARE CRIMINALS AGAINST THE CHILD-THE LOWEST FORM OF HUMAN
So, the cross-reactivity of the quadrivalent vaccine with its “almost unavoidable” autoimmunity wasn’t bad enough? A nonavalent vaccine will be much worse. What’s the real goal of this vaccination campaign? To kill, cripple, and sterilize?
Working with statistics released by the Japanese government, I estimate that with the quadrivalent Gardasil vaccine, 7% of vaccine recipients reported serious adverse effects lasting at least several months after vaccination. This is 1 in 14 people. The serious adverse effects included seizures, disabling headaches, and whole body pain. What we are looking at here is autoimmune disease of the central nervous system, and autoimmune disease similar to rheumatoid arthritis (affecting the whole body).
At least seven distinct mechanisms of harm have already been identified for quadrivalent Gardasil. Gardasil should be taken off the market, not bumped up to an even higher level of harm.
Pathetic as even now my daughter remains disabled after HPV4 Gardasil vaccine since 2007 – now there’s a much higher risk of damage from a 9-Valent HPV vaccine! How crazy is the CDC?! Follow the Money, Honey – let’s see who profits from our vaccine injured kids!
I have also read that the so called safety tests are fake. In producing the first gardasil vaccine, the vaccine was compared to a “placebo” of aluminum. Now, for this second gardasil vaccine, the “placebo” was the first gardasil vaccine.
Please, people, stop poisoning your vulnerable children with ANY vaccine. Do the simple research of reading (online) the actual package inserts telling the INGREDIENTS and SIDE EFFECTS of what these useless, harmful injections do to infants, children and adults. Take back your right to a strong, natural immune system. We have been fooled, brainwashed, injured and killed by this legally indemnified industry for far too long. cheers
This is insane. Let’s give boys a vaccine made for girls, tell the public why boys need it so they’ll comply and see how much more $$$ we can make. Then let’s make a mandate that we aren’t responsible for reactions to our vaccines, this way we have no accountability for our actions. We’ll then blame the parents for their inability to parent and lay the blame at their feet.
All my kids are vaccine free and I’m great with that. I had the Hep B shot for work which paralyzed my right arm for several hours. The nurse told me she must have hit a nerve. (That would have been instant, not gradual like this was). That is when I started to educate myself. I spent hours pouring over evidence of the ill effects, I’ve been publicly and loudly yelled at by a doctor for choosing not to vaccinate. If my arm was paralyzed what would it do to my newborn. I have never withdrawn any of my kids from school from an outbreak, but I gladly would. My oldest was rejected from online public school last year because I didn’t immunize. Why would that matter? He’s at home. Its not going to transfer through the computer. That’s what fear does for you. Why is there a chicken pox vaccine, you get it once and your immune. Why does a newborn need the Hep B shot? Its transmitted theough sexual contact. I could go on and on. I recently told a nurse my child was not going to get the tetanus shot. I was again lectured. People are afraid their child will get tetanus from my child. Please! That’s ridicules. Its like saying my child has a cut on his hand and if he touches your kid he will give the cut to your kid. Its a government power issue that includes $$$$, payouts, and dumbing down society for control. If we can’t fight, they win.
Yes this is insane. Thank you Becky.
Virus cannot, will not and could never cause cancer in humans. This is the lie behind the present inane CDC claim that has its beginnings back in the 70’s during Nixons Cancer-Virus Program. Which many conspiracy researchers were sure was a military program to create a cancer causing virus. Problem was, they didn’t know anything about either cancer or viruses.
OK folks, 2 types of virus that virologists know about and study. DNA and RNA.
DNA virus are HPV, HSV1 and HSV2, shingles, chickenpox etc….that cause a specific sore, rash, wart etc… and kill the host cell. They are cytotoxic. They are easily isolated in the blood or tissue in millions of particles called viremia. They are easy to identify.
RNA virus, also referred to by virologists as passenger, associated, or retro….are harmless. They do not kill the host cell. They are extremely hard to isolate in the blood or tissue. They transport coded information between cells. They are made by our bodies in the 1,000’s every day. In some animals, chickens in particular they are said to cause tumors. Said to. The most often quoted example is the Rous Chicken virus.
RNA virus were studied in Nixons Cancer Virus Program, the thinking was they could, because they didn’t destroy their host cell,be the catalyst for that first cell mutation. But they weren’t, in humans. End of program. Money dried up. CDC looking for other crisis pathogens to hype and receive major funding for the study of….enter HIV – yep an RNA virus.
But why let a little program like the Nixon CVP get in the way of making a name for yourself in the field of virology? Enter Robert-never saw a RNA virus that couldn’t be rehabilitated-Gallo and whala! HIV! HTLV1 and HTLV2! All bull! And many virologist were taken in by the hoax, and still are.
Now enter HPV = Cancer of cervix. Can’t happen. Did you know CDC tried to blame cancer of cervix on genital herpes in the 90’s? Which makes more sense if you were going to lie and blame a DNA virus, since genital herpes is an epidemic! But still, its a DNA virus.
Cancer is irregular unregulated cell growth,aplasia, metastasis.
Opposite of that –
DNA virus is cell death. HPV is a DNA virus. HPV cannot cause cancer.
Who cares? CDC, NIH, NIC, NIAID et. al. don’t. They’re all whores. What do whores do? Anything for money, well lots of money. And that’s what they’re interested in.
STOP VACCINATING YOUR CHILDREN FOR HPV/CERVIX, PENAL, TESTICAL CANCER LIES THEY ARE TELLING !!!!
I am devastated that I didn’t do my research prior to allowing my daughter receive the Gardasil Vax as mandated by RI. She had severe anxiety accompanied by minor vocal and facial tics. After the Vax was given, her vocal, facial and body tics have become so severe, she struggles with ordinary tasks. Life changing event due a totally unnecessary and harmful vaccine. Now they want to make it a nationally mandated vaccine. Criminal is the only word I can say.
hpv vaccines
strains of what kind are in the vaccine
hpv is warts
particles are something that grows over time
10 to 20 years later.
no merck will not, they claim lies
and cannot prove gardasil 9 is safe of not
testing by pharmaceutical for safety
is what is used in courts
merck has not done their job,, pharmaceutical is gone Global attack
HPV Vaccines
Merck : states
LIES LIES LIES
The erased information and the silence of the HPV vaccine syndrome united states of America users of such a vaccine
Merck rights and does not follow their own written material on the approval of such vaccines
Such as:
The first HPV vaccines were approved in 2006, following large clinical trials involving more than 20,000 people. Since then, research covering hundreds of thousands of vaccinations shows a similar safety profile to other childhood vaccines. The safety data for Gardasil 9 is consistent with the HPV4 safety studies.v vaccine
KKrasnowatermnan Gardasil :google search:
2006 in Australia
Those death numbers don’t add
Immediate reactions since in use. Gardasil
Gardasil 9:
Merck shows no authority, Mercks’ Attorney for the MSDS sheets, is not stated on the Material Statistical Data Sheets label, as is required for such approvals, The law cannot prove Gradasil9 is safe or not safe